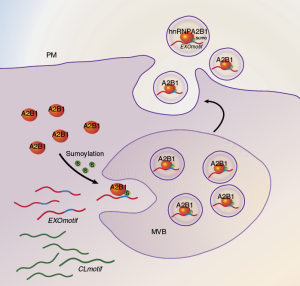
It has long been clear that while many microRNAs are present in the extracellular environment outside of microvesicles, there are many microRNAs that are preferentially packaged into exosomes as they are exported from cells. A major scientific question has been what features of a microRNA cause it to be packaged into microvesicles, and specifically into exosomes. The mechanisms that control the specific loading of RNA species into exosomes have remained obscure. This paper reports on three aspects of one such mechanism. First, a heterogeneous nuclear ribonucleoprotein, hnRNPA2B1, specifically binds exosome-bound miRNAs and directly controls their loading into exosomes. Second, the protein recognizes specific sequence motifs present in the mature miRNA targets and not in other microRNAs. Third, when purified from exosomes, the protein is sumoylated, and sumoylation is required for its binding to export-targeted miRNAs. The authors showed that altering expression level of the protein, its sumoylation level, or presence of the sequence motifs in the target miRNA all changed the level of enrichment of the miRNAs in exosomes versus a control condition. Thus, the authors identified not only the information used in channeling miRNAs into export pathways, but also a protein involved in the targeting and a mechanism (sumoylation) that regulates this process. This appears to be a significant advance in our understanding of the fundamental mechanisms that determine the spectrum and packaging of at least one kind of extracellular RNA.
Villarroya-Beltri, C. et al. Sumoylated hnRNPA2B1 controls the sorting of miRNAs into exosomes through binding to specific motifs. Nat. Commun. (2013) 4: 2980, PMID:24356509, doi:10.1038/ncomms3980.