This blog post comes from the Myotonic Dystrophy Foundation.
Pharmacodynamic Biomarkers and DM
There is now strong support for the concept that a panel of splicing events may serve as a pharmacodynamic biomarker for go/no go decisions in drug development for myotonic dystrophy type 1 (DM1) and Duchenne muscular dystrophy (DMD). Data establishing splicing event sensitivity to free MBNL levels has converged with the natural history of alternative splicing patterns in DM patients to yield a subset of splicing events with the sensitivity and reproducibility to evaluate candidate therapeutics in early stage clinical trials. Quantitative pharmacodynamic biomarkers are invaluable in de-risking industry drug discovery and development, as they facilitate early stage assessment of molecular target engagement and modulation and may inform dose ranging studies. The only caveat is the dependence of these measures upon repeated muscle biopsies (a risk reduced, but not eliminated, by more tolerable needle biopsies). The identification and validation of a non-invasive assay of patient splicing status would be a valuable step forward for clinical trials in DM.
Early Support for a Non-Invasive Biomarker for DM1
Dr. Thurman Wheeler and colleagues at Massachusetts General, Harvard Medical, and Boston Children’s have explored the concept that a subset of extracellular RNAs (exRNAs) released into blood or urine may: (a) reflect alternative splicing status in DM-affected tissues and (b) thereby serve as an easily accessible pharmacodynamic biomarker platform for DM1 (Antoury et al., 2018). These studies were supported in part by a grant to facilitate “Development of Biomarkers for Myotonic Studies” from Myotonic Dystrophy Foundation/Wyck Foundation.
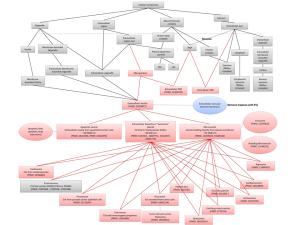
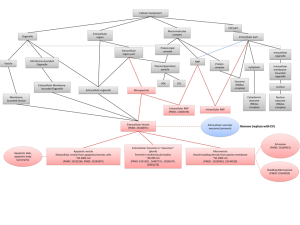
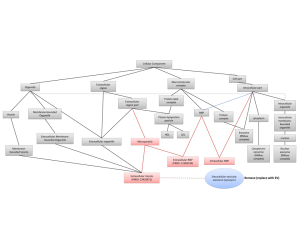
The research team initially found that > 30 transcripts that are alternatively spliced in DM1 muscle biopsies were detectable in human blood and urine samples; follow-up studies confirmed the presence of RNAs in extracellular fluids/exosomal particles. Normalized DMPK expression levels in urine from DM1 patients, by droplet digital PCR, were ~50% of unaffected controls. Assessments of DM1-established alternative splicing events showed that a subset (10/33) also occurred in urine exRNA, including being conserved in longitudinal (6-26 month) studies of the same patients. Assessments of alternative splicing events in blood exRNA did not yield the same value.
Using principal component analysis of 10 alternative splicing events observed in urine exRNA, the research team then generated a putative composite biomarker panel for DM1. The ensuing predictive model of alternative splicing in DM1 proved to be 100% accurate in comparisons of training and independent validation data sets to distinguish DM1 from unaffected controls and in distinguishing disease status of subsequently enrolled subjects. The research team also linked alternative splicing patterns in urine exRNA to variation in DM1 clinical phenotypes, suggesting that modeling of urine exRNA alternative splicing may allow both the tracking of disease progression and the impact of candidate therapeutics.
Finally, to address questions as to the source of urine exRNA, the team assessed alternative splicing in urinary tract cells of DM1 mouse models (the ubiquitous Mbnl1 ko and the tissue-specific HSALR). While kidney and bladder cells of the Mbnl1 ko reflected patterns in skeletal muscle, assessments of the same tissues in the HSALR showed no differences from control mice. These data strongly suggested that the exRNAs assessed in urine reflect exosomes released from urinary tract cells. Some of the alternatively spliced transcripts in urine exRNA also were shown to be altered by antisense oligonucleotide drugs previously shown to correct splicing patterns in DM1 mouse models. The research team’s parallel studies of Duchenne muscular dystrophy also supported the concept that urine exRNA has utility as a pharmacodynamic biomarker in drug intervention studies.
Towards a Non-Invasive Biomarker for DM1
Taken together, these data provide compelling proof of concept that a panel of alternative splicing events assessed in urine may serve as a robust composite biomarker of DM1 progression and as a tool for assessment of candidate therapeutics. A non-invasive biomarker such as this would greatly extend the ability to perform repeated measurements in longitudinal natural history studies (as a disease progression biomarker) and in interventional clinical trials (as patient stratification and pharmacodynamic biomarkers), including making assessment of pediatric DM1 patient cohorts feasible. Although it is not essential to formally qualify a biomarker, existing regulatory agency guidance documents (see References below) provide a valuable evidentiary framework for moving non-invasive biomarker work towards an accepted clinical tool for DM1.
References
Antoury L, Hu N, Balaj L, Das S, Georghiou S, Darras B, Clark T, Breakefield XO, Wheeler TM. Analysis of extracellular mRNA in human urine reveals splice variant biomarkers of muscular dystrophies. Nat Commun. (2018) 9: 3906. doi: 10.1038/s41467-018-06206-0. PMID: 30254196
Framework for Defining Evidentiary Criteria for Biomarker Qualification. Foundation for the National Institutes of Health (FNIH) Evidentiary Criteria Writing Group. October 2016. (announcement) (pdf)
Guidance for Industry and FDA Staff: Qualification Process for Drug Development Tools. (pdf)