This week, the Journal of Extracellular Vesicles has published a special issue focusing on the work of the Extracellular RNA Communication Consortium.
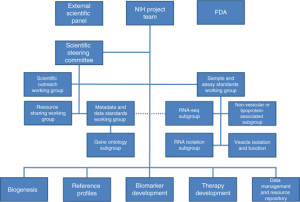
The NIH Common Fund has identified extracellular RNA as a promising new area of biology, and has put together a comprehensive program to explore it. The goal is to understand basic biology, develop new protocols for isolating exRNA, find out the levels of exRNAs in healthy biofluids, and then leverage that new knowledge to develop biomarkers and therapies to deal with a range of diseases. Everything the consortium develops will be made available to the wider scientific community so that we can push the field forward together.
Below are links to the six articles in the special issue:
- The NIH Extracellular RNA Communication Consortium
- Biogenesis, delivery, and function of extracellular RNA
- Extracellular RNAs: development as biomarkers of human disease
- Potential Functional Applications of Extracellular Vesicles: a report by the NIH Common Fund Extracellular RNA Communication Consortium
- Integration of extracellular RNA profiling data using metadata, biomedical ontologies, and Linked Data technologies
- Meeting report: Discussions and preliminary findings on Extracellular RNA measurement methods of laboratories in the NIH Extracellular RNA Communication Consortium