This blog post is adapted from a TGen press release, found here.
Uncovering the genetic makeup of patients using DNA sequencing has in recent years provided physicians and their patients with a greater understanding of how best to diagnose and treat the diseases that plague humanity. This is the essence of precision medicine.
Now, researchers at the Translational Genomics Research Institute (TGen) are showing how an even more detailed genetic analysis using RNA sequencing can vastly enhance that understanding, providing doctors and their patients with more precise tools to target the underlying causes of disease and help recommend the best course of action.
In their review, published recently in the journal Nature Reviews Genetics, TGen scientists highlight the many advantages of using RNA-sequencing in the detection and management of everything from cancer to infectious diseases such as Ebola and the rapidly spreading Zika virus.
RNA’s principal role is to act as a messenger carrying instructions from DNA for the synthesis of proteins. Building on the insights provided by DNA profiling, the analysis of RNA provides an even more precise look at how cells behave and how medicine can intervene when things go wrong.

Dr. Sara Byron
“RNA is a dynamic and diverse biomolecule with an essential role in numerous biological processes,” said Dr. Sara Byron, Research Assistant Professor in TGen’s Center for Translational Innovation and the review’s lead author. “From a molecular diagnostic standpoint, RNA-based measurements have the potential for broad application across diverse areas of human health, including disease diagnosis, prognosis, and therapeutic selection.”
DNA (deoxyribonucleic acid) sequencing spells out — in order — the billions of chemical letters that make up the genes that drive all of our biologic make-up and functions, from hair and eye color to whether an individual may be predisposed to cancer or other diseases.
RNA (ribonucleic acid) sequencing provides information on the genes that are actively being made into RNA in a cell and are important for cell function. While more complex, RNA holds the promise of more precise measurement of the human physical condition.
There are more forms of RNA than of DNA present in the body, explains Dr. Byron. “RNA sequencing provides a deeper view of a patient’s genome, revealing detailed information on the diverse spectrum of RNAs being expressed.”
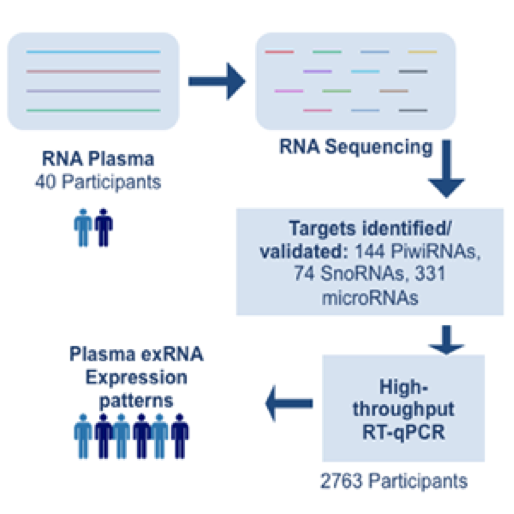
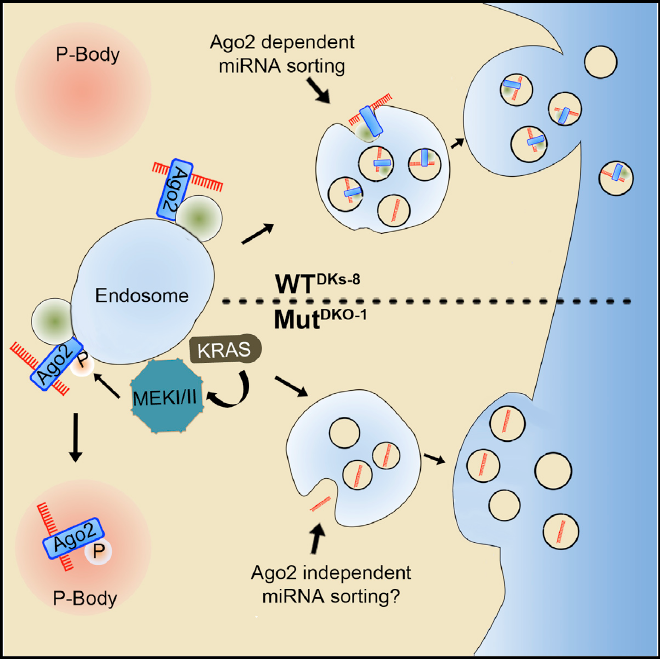
One of the most promising aspects of RNA-based measurements is the potential of using extracellular RNA (exRNAs) as a non-invasive diagnostic indicator of disease. Monitoring exRNA simply takes a blood sample, as opposed to doing a tumor biopsy, which is essentially a minor surgery with greater risks and costs.
“The investigation of exRNAs in biofluids to monitor disease is an area of diagnostic research that is growing rapidly,” said Dr. Kendall Van Keuren-Jensen, TGen Associate Professor of Neurogenomics, Co-Director of TGen’s Center for Noninvasive Diagnostics, and one of the review’s authors. “Measurement of exRNA is appealing as a non-invasive method for monitoring disease. With increased access to biofluids, more frequent sampling can occur over time.”
The first clinical test to measure exRNA was released earlier this year, the review said. The test is for use in evaluating lung cancer progression, and the potential for using RNA-seq in other cancers is expanding rapidly. Commercial RNA-seq tests are now available, providing the opportunity for clinicians to more comprehensively profile cancer and use this information to guide treatment selection for their patients, the review said.
In addition, the authors reported on several recent applications for RNA-seq in the diagnosis and management of infectious diseases, such as monitoring for drug-resistant populations during therapy and tracking the origin and spread of the Ebola virus.
Using examples from discovery and clinical research, the authors also describe how RNA-seq can guide interpretation of genomic DNA sequencing results. The use of integrative sequencing strategies in research studies is growing across a broad range of health applications, which promises to drive the incorporation of RNA-seq into clinical medicine as well, the review said.
The paper, Translating RNA-sequencing into Clinical Diagnostics: Opportunities and Challenges, was published online recently in the journal Nature Reviews Genetics. Authors Kendall Van Keuren-Jensen and David W. Craig are participants in the Extracellular RNA Communication consortium.
Source: Translational Genomics Research Institute