This blog was adapted and simplified from a Spotlight in the Journal of Cell Biology.
Directed movement along a chemical gradient – chemotaxis – is a fundamental behavior of cells in the body during processes like inflammation, embryogenesis, and cancer metastasis. While the intracellular signaling mechanisms underlying chemotaxis have been investigated by many groups, it is not well known how stable gradients of chemoattractant chemicals are generated and maintained outside the cell, especially given the rapid diffusion of chemicals after secretion.
The amoeba Dictyostelium discoideum (fondly known as Dicty) is a model organism often studied since its biology can shed light on that of human cells. Recent work from Kriebel et al. (2018) showed that extracellular vesicles (EVs) are central to the biology of chemotaxis in Dicty by demonstrating that the main chemical Dicty follows during chemotaxis is synthesized within and released from EVs.
Dictyostelia are soil-living amoebas that undergo chemotaxis toward cyclic adenosine monophosphate (cAMP) under starvation conditions. As cAMP is released from the rear of migrating cells, chemotaxis toward cAMP leads to aggregation of the amoebas into multicellular structures.
Kriebel et al. previously showed (2008) that the enzyme that synthesizes cAMP, adenylyl cyclase (ACA), is present in multivesicular bodies (MVBs) located at the rear of migrating amoebas. They have now shown that leader cells release ACA-enzyme-containing vesicular trails and that follower cells stream onto them in a head-to-tail fashion. The vesicular trails are highly chemotactic and direct the migration of the follower cells.
Measurement of ATP, the precursor used by the enzyme to synthesize the product cAMP, revealed that it is also present inside EVs. Narrowing down from the 68 transporter proteins found in Dicty, the researchers identified the one protein – ABCC8 – that transports cAMP from the inside to the outside of EVs in order to promote chemotaxis. Dicty amoebas with that transporter knocked out exhibit much less chemotaxis and streaming behavior, and fluorescently tagged ABCC8 reintroduced into those amoebas is visible in the vesicular trails but not the plasma membrane.
Altogether, these data indicate that the entire machinery for generating and releasing chemoattractants is contained within EVs: the catalytically active enzyme (ACA), its substrate (ATP), the product (cAMP), and the transporter (ABCC8; see figure).
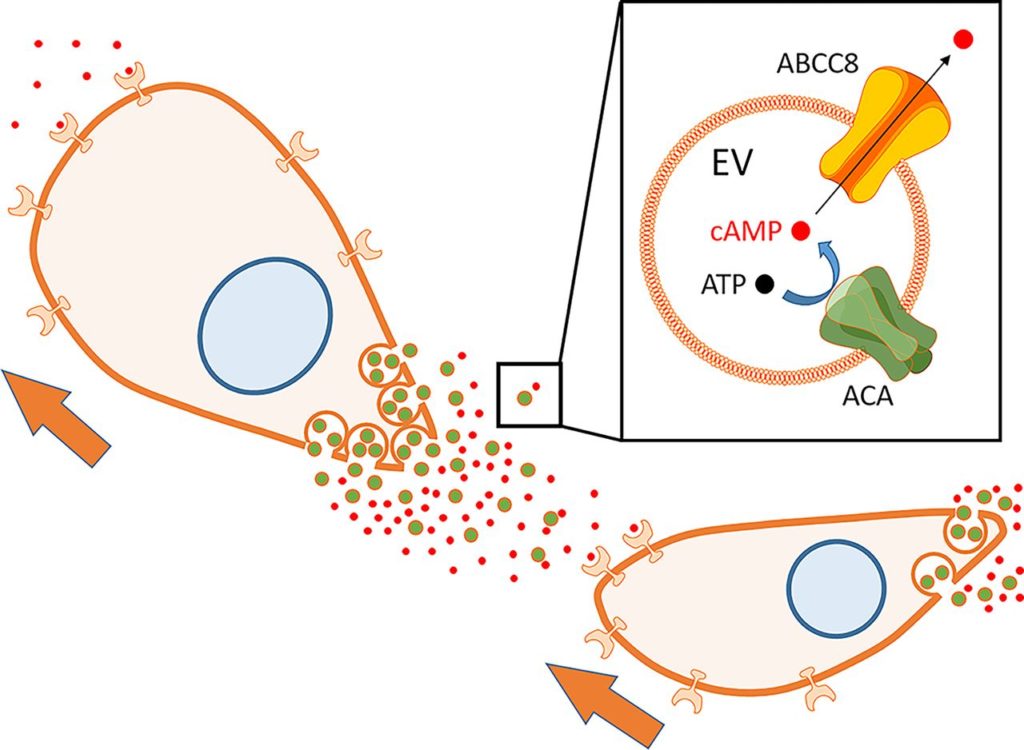
EVs secreted from Dictyostelia synthesize and release cAMP to promote chemotaxis. cAMP is converted from ATP by enzyme ACA in EVs and secreted through the ABCC8 transporter. Secreted cAMP makes a gradient and promotes chemotaxis of follower cells.
Overall, Kriebel et al. (2018) describe an elegant system by which chemotactic signals are generated and sustained to promote streaming in a complex multicellular system, and they elucidate a major mechanism by which cells leave a memory of themselves. EVs are left in a breadcrumb-like trail behind cells and continue sending chemotactic signals to surrounding cells. The finding that EVs act as cell-independent entities to not only carry but also generate bioactive products is important because it shows that they can amplify signals.
The same group made similar findings previously for neutrophils, one of the main sentry cells of the immune system (Majumdar et al., 2016). In both cases, the presence in EVs of enzymes that can generate a chemical product continuously amplifies and sustains a signal beyond that which would occur if the EVs carried just the chemical. The findings in amoeba and human cells together suggest that amplification of chemotactic signals is an important function of EVs that is conserved across organisms and necessary to promote effective directional sensing.
In a different context, it has been reported that precursor miRNAs can be processed into mature miRNAs in exosomes in a cell-independent manner due to the presence of the RNA interference–silencing complex (RISC) machinery in breast cancer–associated exosomes (Melo et al., 2014) and that this activity is important to promote tumor aggressiveness. Along with Melo et al. (2014), Kriebel et al. (2018) provide direct evidence that EVs can act as an independent machinery to regulate biological processes such as chemotaxis or tumorigenesis via enzymatic activities.
References
Kriebel PW, Barr VA, Rericha EC, Zhang G & Parent CA . (2008) Collective cell migration requires vesicular trafficking for chemoattractant delivery at the trailing edge. J. Cell Biol. 183:949–961. doi: 10.1083/jcb.200808105
Kriebel PW, et al. (2018) Extracellular vesicles direct migration by synthesizing and releasing chemotactic signals. J. Cell Biol. doi: 10.1083/jcb.201710170
Majumdar R, Tavakoli Tameh A & Parent CA. (2016) Exosomes mediate LTB4 release during neutrophil chemotaxis. PLoS Biol. 14:e1002336. doi: 10.1371/journal.pbio.1002336
Melo, et al. 2014. Cancer exosomes perform cell-independent microRNA biogenesis and promote tumorigenesis. Cancer Cell 26:707–721. doi: 10.1016/j.ccell.2014.09.005
Sung BH & Weaver AM. (2018) Directed migration: Cells navigate by extracellular vesicles. J. Cell Biol. doi: 10.1083/jcb.201806018

