06.15.16 last updated.
06.15.16 last updated.
Extracellular vesicles (EVs) play an important role in cell-to-cell communication. Recently, EVs have been shown to be involved in immune modulation, tumor biology, and tissue regeneration. The mechanisms of action of EVs are associated with their ability to stimulate target cells directly and to transfer proteins, biologically active lipids, and nucleic acids to the target cells. In fact, mRNAs, long non-coding RNAs (lncRNAs), and microRNAs (miRNAs) can be compartmentalized into EVs, escape enzymatic degradation, and be delivered to target cells. This horizontal transfer of extracellular RNAs carried by EVs can induce epigenetic alterations in recipient cells. The result is a change of phenotype or even a genetic and functional reprogramming of the recipient cells. Furthermore, EVs carry a selection of miRNAs different from the miRNAs most expressed in the cells of origin. However, little is known about the mechanisms of miRNA enrichment in EVs.
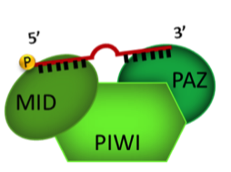
Argonaute2

Alix
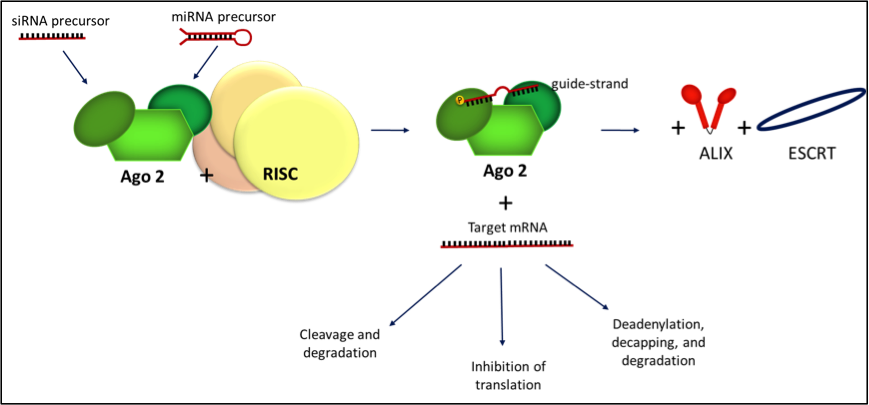
We hypothesized a possible interaction between the Alix and Argonaute 2 (Ago2) proteins. The resulting complex may have a role in miRNA transport into EVs. Ago2 is a reasonable candidate to play a role in miRNA packaging within multivesicular bodies during EV biogenesis because of its central role in miRNA maturation. We observed that Ago2, as well as several other ribonucloproteins involved in RNA storage and stability, is expressed in EVs derived from adult human liver stem-like cells (HLSCs). Cells which express mesenchymal and embryonic markers.

Alix is a multifunctional protein commonly used as a marker of EVs. It is an accessory protein of the Endosomal Sorting Complex Required for Transport (ESCRT), and several studies indicate that ESCRT is involved in the biogenesis of EVs.
We observed that HLSC-derived EVs express both Alix and Ago2. Co-immunoprecipitation (Co-IP) experiments with Alix or Ago2 antibody showed that the two proteins are associated. We also found that the miRNAs enriched in HLSC-EVs precipitate with the Alix – Ago2 complex. After the incubation of HLSC-EVs with human endothelial cells, we observed that miRNAs from HLSC-EVs are transferred to these cells.
After the silencing of Alix expression in HLSCs, we observed the absence of both Alix and Ago2 proteins in EVs derived from the knockdown HLSCs and a strong reduction in the number of miRNAs normally enriched in HLSC-EVs. On the other hand, EV size, surface expression of CD63 and Tsg101, and the number of released EVs were not affected. After incubation with endothelial cells, EVs derived from Alix-knockdown HLSC do not transfer miRNAs to cells.
Alix is known to be involved in endocytic membrane trafficking and cytoskeletal remodeling. It is also associated with the ESCRT machinery, which participates in processes of vesiculation and cargo sorting, including multivesicular body biogenesis. Our data suggest that Alix binds Ago2 and drives it into EVs together with the associated miRNAs.
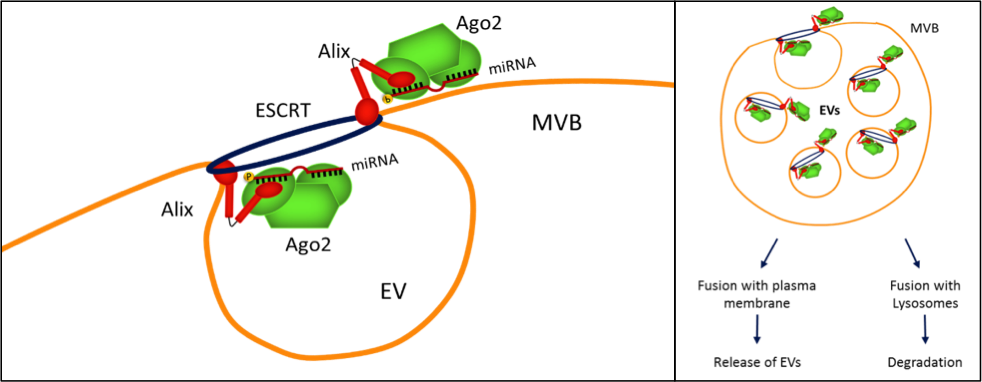
This might be a general mechanism of miRNA transport into EVs, common to other cell types. Enrichment of a selected set of miRNAs might also depend on the affinity of miRNAs for carrier proteins such as Ago2.
Source: Iavello A, Frech VS, Gai C, Deregibus MC, Quesenberry PJ, Camussi G. Role of Alix in miRNA packaging during extracellular vesicle biogenesis. Int J Mol Med. 2016 37:958-966. doi: 10.3892/ijmm.2016.2488. PMID: 26935291.
Hypoxia promotes tumorigenesis by downregulating miRNAs
Protocols for studying extracellular vesicles and extracellular RNA
There are no comments.